The Anatomy of a Clinical Trial
Clinical trials are a complicated mess, find out why and what is being done to fix them
Welcome to all the new subscribers that have joined us over the last few weeks! If you haven’t already, you can read my last article on Startup Moats and Defensibility here.
Stay up to date with Superfluid by subscribing here:
Over the last few weeks, I've been writing about drug discovery and the potential AI has in accelerating workflows and driving efficiencies at the pre-clinical stage. As a quick refresher, the pre-clinical stage refers to lead identification, compound screening and in vivo/in vitro testing.
While there is a ton of uncertainty at the pre-clinical stage, much of this clears up as a drug candidate goes through clinical trials. You find out if your drug actually works in humans or not and whether there is a requisite level of efficacy for it to be commercially viable.
This stage is also by far the most expensive. It costs hundreds of millions of dollars to run a clinical trial, and for every day it is delayed, it can result in US$600k - 8m in losses for a trial sponsor.
Mind-blowing! 🤯
With that data point, it's clear that improving the success rate of drugs that go through clinical trials is a ridiculously high-value exercise to pursue. This can happen in the pre-clinical stage (what we've covered to date), but also through the clinical trial process, which is what we'll be covering today.
Clinical Trials 101
The goal of a clinical trial is to augment the current medical knowledge base and bring novel treatments to patients in a safe and regulated manner. Clinical trials typically follow a 3 phase process, followed by a maintenance stage.
Phase 1: The first phase usually involves ~20 or so healthy patients with the goal of determining the drug's safety profile, dosage level and side effects. Typically pharmacokinetic modelling and simulation are used to optimise dosages and minimise adverse reactions. This phase is the first time the new drug is tested in a human body and provides researchers with valuable insight into how the drug interacts with the body.
Phase 2: With the drug's baseline dosage and safety validated in Phase 1, we now move on to a larger patient group (several hundred) that have the target disease. The goal for this phase is to evaluate the drug's efficacy and to further identify any safety considerations in a larger patient population. At this stage, trial design is incredibly important to ensure that the results obtained are both statistically significant and clinically relevant. This often involves the use of control groups, randomisation, and blinding techniques to minimise bias and confounding variables. As the trial progresses, data is collected and analysed to determine the drug's optimal dosage, therapeutic effects, and potential side effects.
Phase 3: This is the most critical stage of the clinical trial process where a larger patient group is used to fully confirm the drug's efficacy and safety in a real-world environment. The patient group is typically split up across multiple sites globally to replicate different demographics and characteristics. At this stage, the drug also needs to be able to demonstrate its superiority over existing treatments. Further data is collected at this stage to identify safety signals and optimise treatment strategies.
The maintenance phase: Post-release, the safety and efficacy of the drug is monitored in the general population to detect any new safety concerns or side effects that may not have been identified through the clinical trial process.
To be able to execute this multi-stage process, there are numerous stakeholders that all play important roles to be able to bring a drug to market.
Study Participants: Clinical trials obviously wouldn't be possible without patients who take part and provide valuable data on the safety and efficacy of new treatments.
Principal Investigator: The lead investigator is responsible for the overall conduct of the clinical trial at a specific site. They are most relevant during the study design, implementation, and data analysis stages, as they lead the research team and ensure the trial is conducted according to protocol.
Sponsor: The sponsor funds the clinical trial and is responsible for its overall management. This is typically the biotech/therapeutics business that developed the drug initially but can be other organisations such as an NFP, government department or university. The sponsor designs the trial protocol, obtains regulatory approval, provides funding, and oversees the conduct of the study. They are also responsible for ensuring the safety and well-being of the study participants and for the accuracy and integrity of the trial data.
Contract Research Organisation (CRO): CROs are responsible for managing various aspects of clinical trials on behalf of a sponsor. They typically handle site selection and management, patient recruitment, data collection, analysis and quality control. CROs can also provide expertise in study design, protocol development and regulatory compliance to ensure that the trial is conducted ethically and accurately.
Regulatory Authorities: These are organisations like the Therapeutic Goods Administration in Australia or the US Food and Drug Administration which oversee clinical trials to ensure that they are compliant with federal laws and regulations. They are responsible for reviewing and approving trial protocols and conducting and evaluating the trial's results prior to approving a new treatment
IRB/IEC Ethics Committees: These committees ensure that the patient’s rights and welfare are protected during a clinical trial. The committee has oversight over study design and implementation as they review and approve trial protocols, monitor trial conduct and ensure all patients are fully informed through the trial.
Navigating the Challenges of Running a Clinical Trial
Patient Recruitment and Enrolment
The biggest and costliest part of the clinical trials process is managing patient recruitment, enrolment and participation. Depending on the disease and the parameters of the trial, this step of the process can be drawn out for months or even years. In general, for rare diseases or specific patient populations, it might be hard to find people with that disease (i.e., only 1 in 100,000 people might have the disease), they might be spread globally making it difficult to identify, recruit and retain them for a trial and there might even be reluctance from the patient due to the experimental nature of the treatment. Additionally, many clinical trials often have strict inclusion and exclusion criteria to ensure patient safety and reliability of results which further limits the available pool of participants.
For example, Pfizer recently removed 50% of their Phase 3 Lyme disease vaccine patients due to Good Clinical Practice violations at some trial sites. As a result, the trial has been delayed significantly with additional recruitment costs to be incurred. Furthermore, the FDA could react negatively and request further studies which will delay the trial further.
Intense competition also exists among multiple trials targeting the same patient population. This means that it is crucial for the right patients to be aware of all the ongoing trials which is a large problem that multiple well-funded startups are trying to solve at the moment.
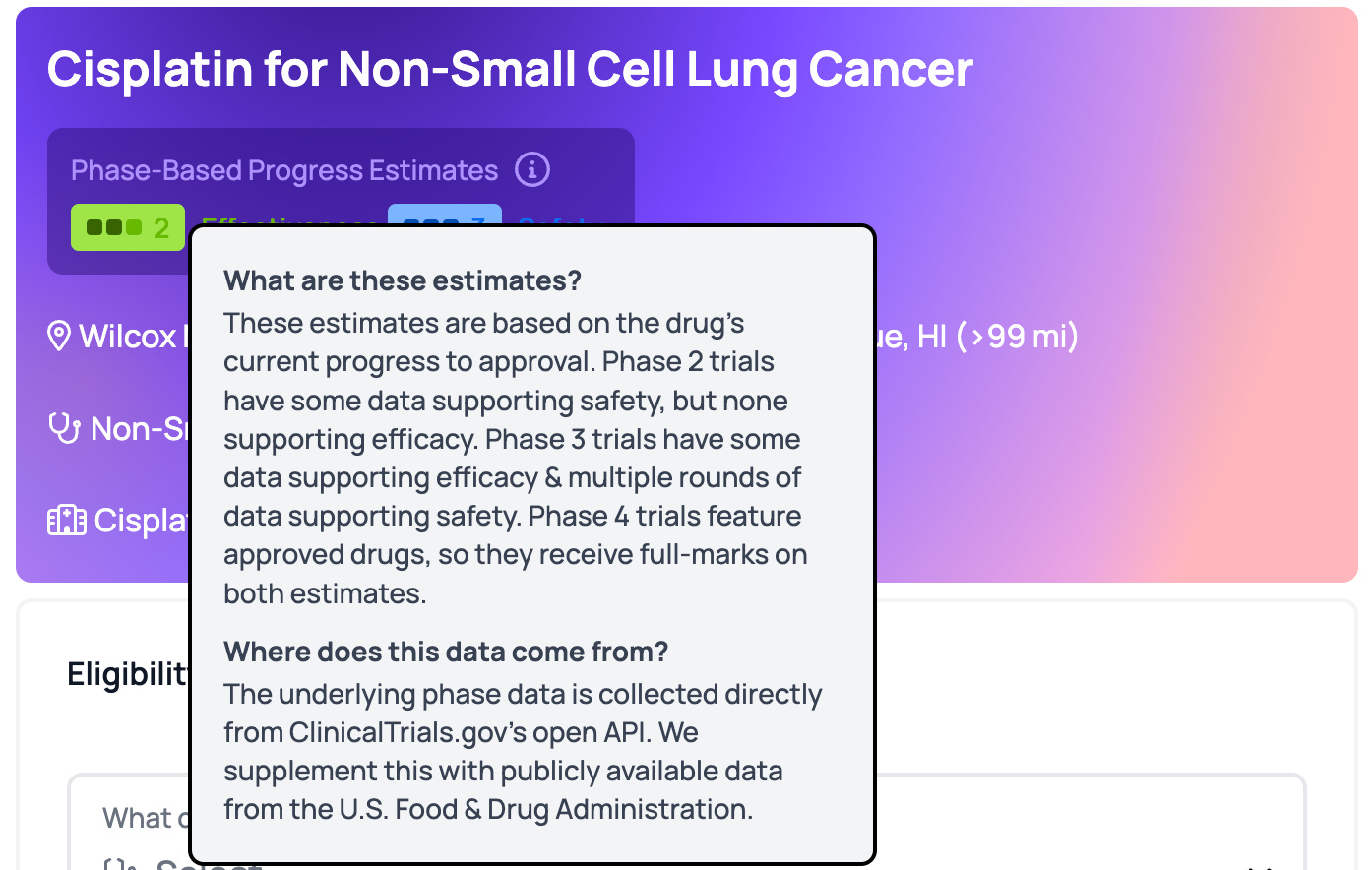
Power is one such startup, and is a clinical trial marketplace that aims to make it easier for patients to identify and sign up for clinical trials. The focus for the marketplace is to give greater look through different trials by providing 'Effectiveness' and 'Safety' scores for each trial as well as looking through trial design and study objectives, giving clarity to patients so that they can make an informed choice before signing up to the trial.
By giving patients more information upfront, Power is likely looking to reduce mid-trial dropout rates and source 'qualified' patient leads for CROs. Power also places a heavy emphasis on UX and reducing the barriers to entry for patients to actually apply for a clinical trial. The process takes under 5 minutes, and the application is also scaled across other relevant clinical trials (see image below). Whilst it's unclear how Power is performing revenue-wise, I suspect they get a finders' fee for each lead sourced and placed. Given this is a costly process for a CRO, you could imagine, each lead costing anywhere from a few thousand dollars to tens of thousands of dollars depending on the eligibility criteria.
Evrima is another targetting the recruitment space and is women-led out of Australia. Evrima takes a different approach towards patient recruitment by focusing on the connection between clinical research and primary care by integrating clinical trial information into electronic health records (EHR). Evrima uses the EHR to determine whether a patient is eligible for a trial based on its inclusion and exclusion criteria. This means that GPs are empowered to access trial opportunities for their patients seamlessly and cut down on the administrative burden faced by patients when signing up for a trial.
Clinical Trial Logistics
Whilst phase 1 trials are fairly straightforward to run, Phase 2 and 3 become complicated with different testing procedures, larger patient pools and data collection needs. On the logistics side, dealing with the hassles of multiple trial sites, temperature and time-sensitive materials is a nightmare for CROs to contend with. The ability to minimise the costs associated with the transportation of clinical goods globally as well as ensuring that these items are kept secure within complex supply chain networks is a paramount problem to be solved.
Slope is a clinical operations startup that provides research sites and CROs with supply chain management software that is purpose-built for sample-intensive and complex clinical trials. Slope becomes the clinical source of truth by configuring protocol-specific workflows and processes for supply procurement, inventory management, biological sample collection and other procedures. Slope then takes a data-driven approach to gauge supply demands by using patient-level data to orchestrate the operational execution of a clinical trial.
In the case of this clinical trial, Slope tracked the clinical inventory needed for the trial at all sites and their status. The platform also ensures the demand is tracked accurately so that there is never a shortage of lab kits but also so that there is less wastage and higher supply utilisation. This significantly drives down the cost of running a clinical trial and in this case, saved the trial ~US$50k+ on just clinical supplies alone, not to mention over US$250k on other operational improvements. Keep in mind that this was only a Phase 1 trial with 30 participants, so you can clearly see that if this was multiplied by a factor of 10+ for later trials the savings are quite substantial.
Data Analytics
As you might already be able to gather, clinical trials generate massive amounts of data from numerous sources including patient records, medical devices and lab results which all come in different forms and formats. This makes it difficult to clean, manage and analyse.
Clinical trials involve participants with different genetic backgrounds, environments, and lifestyles, and they usually measure multiple outcomes and biomarkers to assess how effective and safe treatment is. However, missing data and people dropping out of the trials are common, which can lead to biased results if not dealt with properly. This is why advanced statistical techniques, like multiple imputation or mixed-effects models, are needed to accurately analyse the data and draw meaningful insights.
There are numerous companies dealing with clinical trial data, but I wanted to focus on an interesting company called Unlearn.AI. A necessary evil of clinical trials is to place patients within a placebo group. Keep in mind these are patients that are currently experiencing the disease that the drug is hoping to fix, but are given placebos despite thinking they are receiving a (sometimes) life-saving treatment.
Placebos are necessary to validate and verify the efficacy of an experimental drug, however, can be construed as unethical to the patient. Unlearn is hoping to remove the need to have a control group by effectively creating a digital twin of any patient within a study. Unlearn's team invented their own statistical modelling approach called a Boltzmann-Encoded Adversarial Machine (BEAM) that receives patient data at the start of a trial, generates a digital twin and predicts how they would trend if they were given a placebo.
BEAM is trained on placebo data from prior trials and the outputs are supposedly statistically indistinguishable from the biomarkers of real placebo patients. Whilst the goal is to eradicate placebo groups entirely, at the moment, Unlearn serves as an extension to a smaller control group, meaning that there is less time spent at the recruitment and enrolment stage creating a huge cost saving for a trial and accelerating time-to-market for the drug.
The Future of Clinical Trials
Trying to solve the inefficiencies of a clinical trial is nothing new. There have been hundreds of startups tackling this problem and I'm sure there will be hundreds more to follow. So if the problems are clear and the solutions are clear, why hasn't this been solved yet?
Whilst in some cases, especially around the recruitment side, there have been good levels of traction, clinical trials face immense regulatory pressures and stakeholders prefer to reduce the number of risks taken with the process. As a result, this puts new technological shifts on the back burner and requires higher-level executives to proactively seek out solutions to their problems.
In a recent write-up by the New York Times, Daniel Skovronsky, Eli Lilly’s chief scientific and medical officer said that “Speed [is their] No. 1 incentive" after an overhaul of their labourious internal approvals process as a result of numerous misses through the 2010s. As a result of this overhaul, they have managed to bring forth multiple monumental and record-breaking drugs to market including Mounjaro, a weight-loss drug, similar to Ozempic and Wegovy.
By actively abandoning a rigorous approvals process through multiple internal committees and bringing forward clinical trials, Eli Lilly was able to bring forward a drug that cannibalised the sales of an existing drug but was able to serve the end patient better. As a result, Mounjaro could be one of the highest-selling drugs of all time with annual sales exceeding $25 billion which is just insane!
Ideally, this case serves as a waking-up call for the rest of the ecosystem about the benefits of moving faster and discarding previously held business-centric incentives to focus on speed to market and the benefits of using technology to achieve this.
Make sure to subscribe now to not miss the next article.
How did you like today’s article? Your feedback helps me make this amazing.
Thanks for reading and see you next time!
Abhi




Hi Abhishek! As a newbie to the healthtech VC space, there's a lot that I need to quickly catch up on and this was a great read. So please continue posting more such Healthtech 101 articles for people like me.
Great read!